Looking To the East for Clues To Inhibit Prostate Cancer Metastasis

Knowing this information, Raymond Bergan, MD, from Northwestern University began to investigate soy, to see if it could potentially be used to prevent prostate cancer from spreading, to prevent metastasis.
Genistein

A component in soy that appears to have major effects on prostate cancer cells is called genistein.
Dr. Bergan started testing genistein on prostate cancer cells in the lab.
“I wanted to see if cancer cell detachment, the first step in metastasis when a cancer cell separates from its parent tumor, could potentially be affected by genistein,” Bergan said.
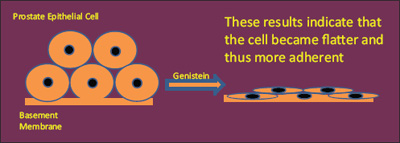
One way of preventing metastasis is to discourage the detachment by making cells more adherent. When cells adhere, they flatten or stretch out.
He found that genistein changes the shape of the prostate cancer cell, in effect causing the cells to hunker down and squash together. Cells in this shape cannot easily move or spread.
“I found that genistein targets and prevents vital steps that must occur for a cell to spread,” Bergan said.
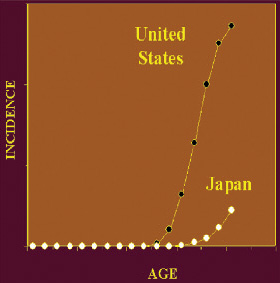
In the next stage, Dr. Bergan injected cells into the prostates of mice to purposefully mimic an aggressive prostate cancer, which grows inside the prostate gland and then spreads outside – to the bones and lungs for example. He gave some of the mice genistein in their food, and discovered that the mice that received genistein had significantly fewer metastases than those who did not receive it.
First Clinical Trials Show Promise
Next Dr. Bergan, in collaboration with Dr. Catalona, moved this research out of the lab and into clinical trials on patients.
To date, they demonstrated that genistein is well tolerated with few and minor side effects (a mild rash at most). Also, the genistein is having similar effects, to those seen in mice, on the prostate cells in humans who have taken genistein 2-3 weeks before they undergo radical prostatectomy for prostate cancer.
“What we found in this small sampling of patients undergoing radical prostatectomy is that this well tolerated soy derivative may represent a potential preventative agent against metastatic prostate cancer,” Bergan said.
Now, in a more recent clinical trial, Drs. Bergan and Catalona are looking at the effects of genistein on highly aggressive prostate cancer.
Men with high-grade prostate cancers, who agree to be part of the trial, have a blood test to detect circulating prostate cancer cells. These cells circulate in the bloodstream looking for a place to rest and begin growing into tumors themselves – similar to throwing grass seed onto moist soil.
Choosing the Patients
Dr. Bergan developed a technique for detecting these cells in a quick and efficient manner. If patients have these cells, they become eligible for the trial and are randomly selected to take a pill for two weeks before their surgery, and three months after. This pill is either a placebo or genistein.
The patients also have a blood sample taken on the morning of their surgery, at three months, and again a year after the surgery to look for circulating tumor cells.
The hope is that the genistein will lower or prevent these tumor cells, which means men will be able to take a dietary supplement to prevent prostate cancer cells from spreading. This trial has just begun but results should be complete in two years.
Too Early for Recommendations
Bergan wants to emphasize that genistein is not available to the public. It is created synthetically for experimental use. And the trials are still in the experimental stage.
The experiment is being done in the context of a research setting where people can be closely monitored by researchers.
“Even though our findings to date look promising, the fact is we have not yet shown that the drug will definitively stop the spread of prostate cancer. If people start taking high doses of every drug that is being tested, they are risking side effects with little chance of benefit,” Bergan said.
Their research suggests that eating soy foods is good.
“However,” Bergan said, “there’s a big difference between amounts of soy in a diet and the higher amounts in concentrated supplements. At this time, I do not recommend soy supplements and, in fact, I would clearly discourage them.”
The good news is the time frame.
“If the trials confirm the effectiveness of genistein, treatment recommendations should be available in two years,” Bergan said.
based upon presentation at May, 2011 AUA meeting: Genistein Inhibits Prostate Cancer Metastasis By Preventing Cell Detachment Barry B. McGuire, MD; Xiaoke Huang, PhD; Irene Ogden, PhD; Daniel C. O’Brien; Phillip R. Cooper; Jessica A. Banks; William J. Catalona, MD; Raymond C. Bergan, MD
Don’t Forget the Technology
Getting helpful information for medical diagnosis and treatment is more than developing a study and reporting results.
Technology has to meet the needs and goals of the study.
A novel technology, Immunomagnetic Enrichment Method, was developed by the Northwestern team to investigate the anti-metastatic action of the soy protein, genistein.
In order to metastasize, cancer cells must invade through the basement membrane into the bloodstream to become a circulating tumor cell or CTC.
The detection of CTCs prior to and during therapy can predict future behavior of the cancer as well as provide targets for further therapy.
With a 24-hour turnaround time in patients with localized prostate cancer, this new test can detect 1 prostate cancer cell in 1 milliliter of blood.
This test is being used in the treatment portion of the clinical phase 2 trial of genistein.
We developed a technique to identify CTCs in patients participating in the Genistein Clinical Phase II Trial. 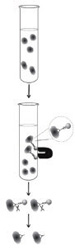
Using antibodies bound to magnetic beads, we isolated CTCs from blood samples.
RNA is purified from the CTCs.
Using this method, we can detect a single prostate cancer cell.
based upon presentation at May, 2011 AUA meeting: Detecting Circulating Tumor Cells in Prostate Cancer Patients: Novel Immunomagnetic Enrichment Method Xiaoke Huang, PhD; Barry B. McGuire, MD;, Irene Ogden, PhD; Mark A. Sparrow; Li Xu; Daniel C. O’Brien; William J. Catalona, MD; Raymond C. Bergan, MD